YOU can advance research towards ReNU treatment options!
📌 Before enrolling in research, from ANY country, Create a Clinical Research ID (CRID)! This is crucial to using the research data collected across platforms and research programs. Once you create a CRID, share it with each clinical research study that you enroll in.
Example for CRID registration: Disease Name: ReNU Syndrome | Gene: RNU4-2 | Variant: n.64_65insT (or your variant)
If there was a treatment for your family member with ReNU syndrome, what would you want it to address? Your voice and perspective matters! This poll helps guide us towards identifying clinical endpoints for future potential treatments for ReNU.
Research Opportunities for Families
Citizen Health
-
Securely organize medical records into a private, digital profile—no site visits required—get instant answers based on YOUR personal journey, and help power research that could lead to new treatments.
Your real-world experiences could contribute to critical natural history studies, enabling researchers to better understand ReNU, uncover patterns, and accelerate progress toward breakthroughs.

Investigation of Newly Discovered Etiologies of Exceptional Disorders
Enroll - email zafiirah.baurhoo@mssm.edu-
If you suspect a family member might have ReNU, you may be eligible for FREE testing. If your family member already has a clinically confirmed genetic diagnosis of ReNU / RNU4-2 or RNU2-2 related disorder and you are interested in participating in the INDEED study, please request a clinical appointment with Dr. Barbosa via MSSMClinicalGenetics@mssm.edu.
Drs. Ernest Turro and Mafalda Barbosa at the Icahn School of Medicine at Mount Sinai have established the INDEED research study to investigate ReNU and other newly discovered genetic conditions. Dr. Turro leads one of the research teams that discovered that mutations in RNU4-2 cause ReNU (Greene et al. 2024). His group also discovered that mutations in a closely related gene, RNU2-2, cause a similar neurodevelopmental disorder (Greene et al. 2025). Dr. Barbosa has over 15 years of experience as a clinical geneticist with a focus on diagnosis and medical management of patients with rare neurodevelopmental disorders. The INDEED study offers free research-use DNA sequencing of the RNU4-2 and RNU2-2 genes.

Clinical Health Survey
Enroll - email NGHI@northwell.edu-
1. Email research team (available Monday-Friday, 9am-5pm ET to answer questions).
2. Receive electronic consent form; sign & return, receive survey link
3. Survey takes ~45 minutes to complete. Questions focus on pregnancy history, genetic testing, medical history (including a brief review of each body system), and family history.
OVERVIEW:
Survey is led by Dr. Ian Krantz, Kelsey Crocker, and Asbaa Khan with the support and expertise of ReNU Syndrome United board members and advisors.
Goal: Perform a cross-sectional study by collecting and analyzing information on the spectrum of features that individuals with ReNU syndrome experience and how these features change or progress over time. Understanding the spectrum of symptoms and their progression empowers parents and guardians to provide optimal care.
Purpose: This study will contribute valuable data to researchers investigating and developing treatments for this condition. Furthermore, it will enhance clinicians' and genetic counselors' understanding of this novel diagnosis, improving recognition of the characteristic features of ReNU syndrome.
Rare-X Registry
-
Rare-X is a critical global registry and ALL are welcome to participate.
Developing treatment options requires that we have robust data that is continually updated.
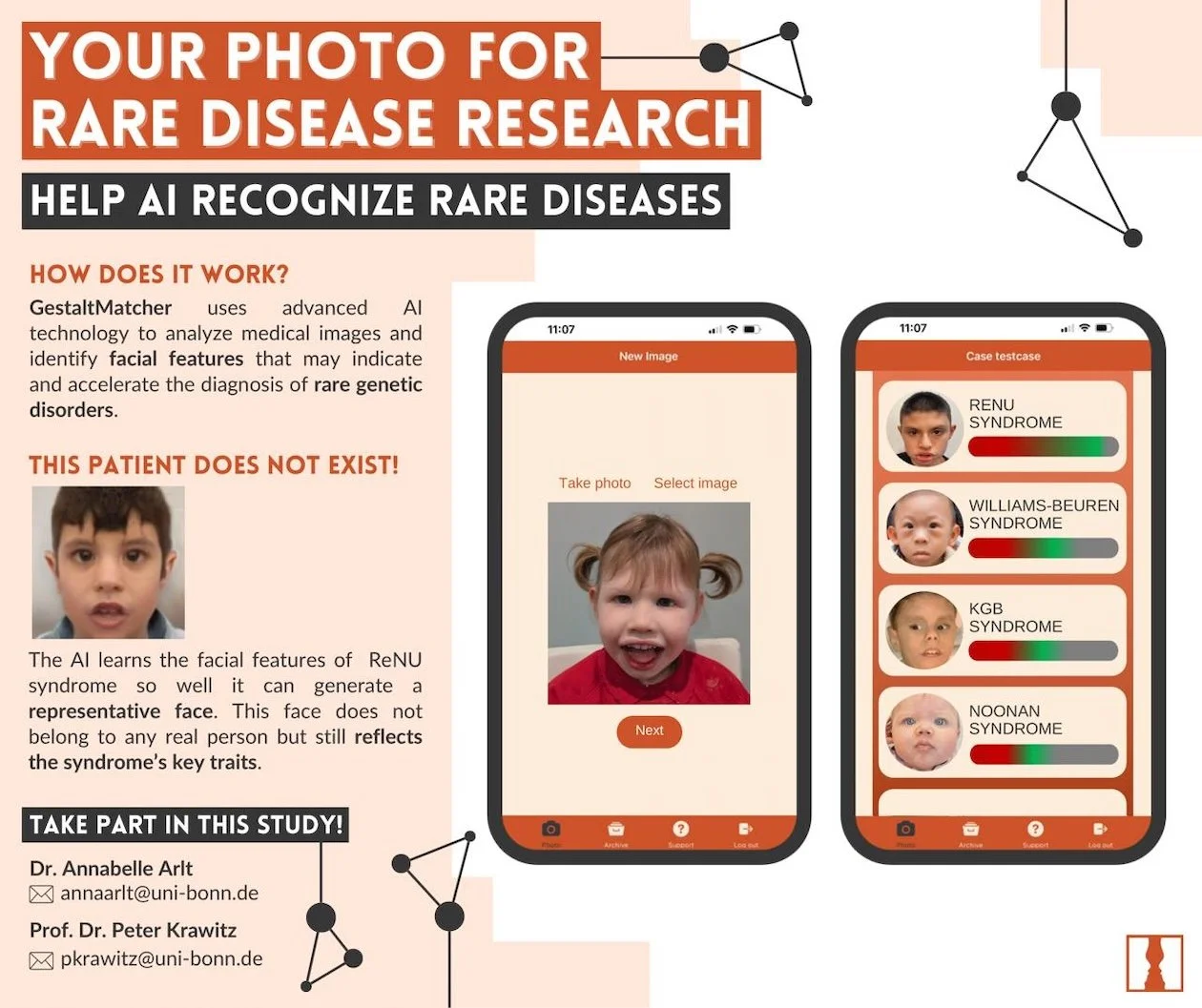
GestaltMatcher
Enroll - email annaarlt@uni-bonn.de-
Contact Dr. Annabelle Arlt or Dr. Peter Krawitz (pkrawitz@uni-bonn.de )
GestaltMatcher uses advanced 2D Al technology to analyze medical images and identify facial features that may indicate and accelerate the diagnosis of rare genetic disorders.

FaceBase
Enroll - email facebase@ucalgary.ca-
3D International Facial Recognition Study. Participation requires <20 minutes. More information at https://hallgrimssonlab.ca/.
The FaceBase team from the University of Calgary is conducting an international study aiming to better characterize genetic syndromes that include facial effects. This includes a computer-based tool using facial imaging to assist physicians in diagnosing syndromes and assessing severity to identify the likelihood of disease-related complications.
Many patients wait years for a definitive syndrome diagnosis or never receive one. Even with a diagnosis, understanding severity and the likelihood of disease-related complications is crucial for clinical decision-making. This may arise due to the absence of known molecular markers for their condition, the presence of an unidentified mutation, or unclear clinical indications for genetic investigation. The FaceBase project aims to bridge this critical gap by enabling the rapid and cost-effective prediction of genotype and associated condition severity through a 3D image of a patient's face. During study participation, video recording is used to minimize challenges that can arise from movement and sensitivities during flash photography. This video is used to select the best moment/image to generate a 3D picture of the participant.
PAID REN Research Study on Rare Epilepsy Experiences
-
Participate in a research study examining the experiences of individuals affected by rare epilepsy syndromes, conducted by the Rare Epilepsy Network (REN).
WHO CAN PARTICIPATE?
This study welcomes:
• Patients (age 18+) diagnosed with a rare epilepsy syndrome
• Caregivers of someone with a rare epilepsy syndrome
• Representatives of epilepsy patient advocacy organizations
• Healthcare providers or researchers in the rare epilepsy field
WHAT'S INVOLVED?
You'll be asked to complete an online baseline survey (40–60 minutes), with optional follow-up surveys every 4–6 months over the course of a year (30–40 minutes each). Surveys cover topics such as disease experiences, treatment history, quality of life, healthcare access, and research priorities — with questions tailored to your participant type.
COMPENSATION
Participants will receive a $100 Amazon gift card for each completed survey, delivered by email within one week of completion.
YOUR PRIVACY
All responses are collected through a secure, encrypted platform. Your name will never be directly linked to your responses, and any findings shared publicly will be fully de-identified.
Participation is entirely voluntary. You may skip any questions or withdraw at any time without penalty, and you will still be compensated for any surveys already completed.
Your input will directly help shape our understanding of rare epilepsy care, identify unmet needs, and guide future research and advocacy efforts.
To learn more or get started, please click here (LINK TO SURVEY).
Frequently Asked Questions
-
This is crucial to using the research data collected across platforms and research programs. When you share it with a research study, it will be used to anonymize your ReNU child, so that researchers may learn from the data you share: they associate your child with a clinical research ID instead of getting names or personally identifiable information about them. If you enroll in multiple research studies, the hope is that by cross-referencing your CRID, you may not need to repeatedly answer the same questions you’ve already answered in previous studies. As an organization, we are encouraging lots of research into ReNU so that it can fuel the development of potential treatment options.
-
Registries are organized systems to collect and store standardized data about RNU4-2 / ReNU Syndrome individuals, enabling researchers to study treatment options, disease progression, and other factors related to healthcare over time.
-
A natural history study is research that tracks how a disease develops and changes over time—without testing a new treatment. It is the foundation of all effective research by turning scattered patient experiences into structured knowledge that makes treatments possible.
It answers: What happens when this condition runs its natural course?
Researchers collect real-world data like:
Symptoms and how they evolve
Age of onset and progression patterns
Lab results, imaging, and medical history
Differences between individuals
Why it’s important
1. Establishes a baseline
You can’t measure if a treatment works unless you know what “normal progression” looks like.2. Identifies meaningful outcomes
It helps define what to measure in clinical trials (e.g., mobility, cognition, biomarkers).3. Reveals patterns and variability
Shows how the condition differs across patients—critical for rare diseases like ReNU Syndrome.4. Accelerates drug development
Regulators (like the FDA) often require natural history data to interpret trial results.5. Improves patient care now
Even before treatments exist, it informs better monitoring, earlier diagnosis, and care planning. -
When you register here on our RNU4-2 map, there is a check-box to opt in to be contacted for future research opportunities.
ReNU Syndrome United is proud to serve as a conduit to connect families with research opportunities. Participation in any study is voluntary, and individuals should carefully review all consent forms and study details before enrolling. All participation is at the individual/family’s own risk.

Exciting research is happening! Zebrafish (center) are used to study ReNU syndrome, for example examining brain changes (left) and behavior differences (right), providing insights into the disease.
To get meaningful data about the progression of RNU4-2 / ReNU syndrome, we need you to join Citizen Health!
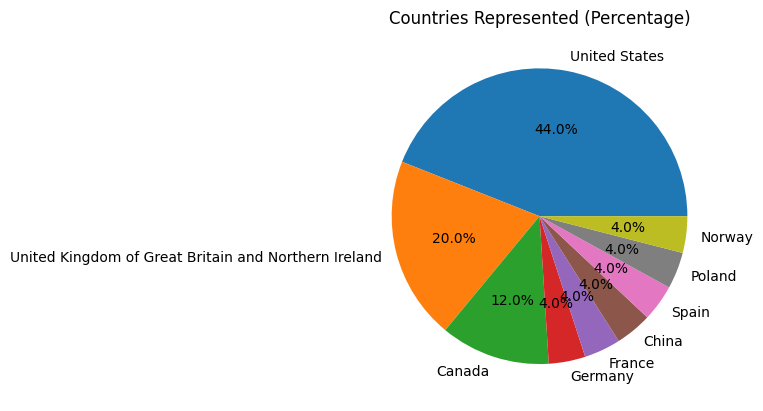
Snapshot of registrants as of January 26, 2026:
What’s important to the RNU4-2 Community?
Take the Treatment Priority Poll
Initial analysis 26 January 2026, re-analysis 3 April 2026
Top 10 priorities in rank order, based on the weighted caregiver poll results:
Absent / delayed speech & communication
Epilepsy / seizures
Brain abnormalities
Mobility
Low muscle tone (hypotonia) ⬆️ (moved up from #6)
Feeding ⬇️ (shifted down one)
Bone fragility
Drooling
Lower gastrointestinal issues (e.g., constipation)
Short stature / poor growth 🆕 (replaces sleep disturbances)